Immunotherapy Increases the Cost of Cancer Care but Reduces Mortality
Immune checkpoint inhibitors (ICIs) are immunotherapy drugs that mobilize the patient’s immune system to detect and attack cancer cells. They are considered a breakthrough development in cancer care, but are very expensive, with a full course of treatment costing more than $150,000 per patient. In The Impact of Immunotherapy on Reductions in Cancer Mortality: Evidence from Medicare (NBER Working Paper 34317), Danea Horn, Abby E. Alpert, Mark Duggan, and Mireille Jacobson use Medicare claims data to evaluate the impact of the first ICIs on healthcare use, costs, and mortality among beneficiaries diagnosed with metastatic melanoma.
Total treatment costs for melanoma patients who used immune checkpoint inhibitors were $85,000 higher than for those who did not, while one-year mortality was 28 percent lower.
The Food and Drug Administration (FDA) first approved ICIs to treat metastatic melanoma; ICIs have subsequently been approved to treat more than 27 types of cancer. The FDA approved the first ICI in 2011 and two additional ICIs in 2014. This innovation in melanoma care was considered particularly important because traditional treatment options, such as radiation therapy and chemotherapy, had limited efficacy against melanoma.
To estimate the impact of ICIs on melanoma patients, the researchers compare changes in patients’ outcomes after the introduction of ICIs to changes over the same period for a control group of patients with metastatic colorectal cancer. ICIs were not approved for the treatment of colorectal cancer until 2017. They are indicated for all metastatic melanoma patients, but only for the approximately 15 percent of metastatic colorectal cancer patients with specific biomarkers. Colorectal cancer and melanoma affect fee-for-service Medicare patients of similar age, with similarly high baseline mortality rates and, prior to the approval of ICIs, similarly trending treatment costs.
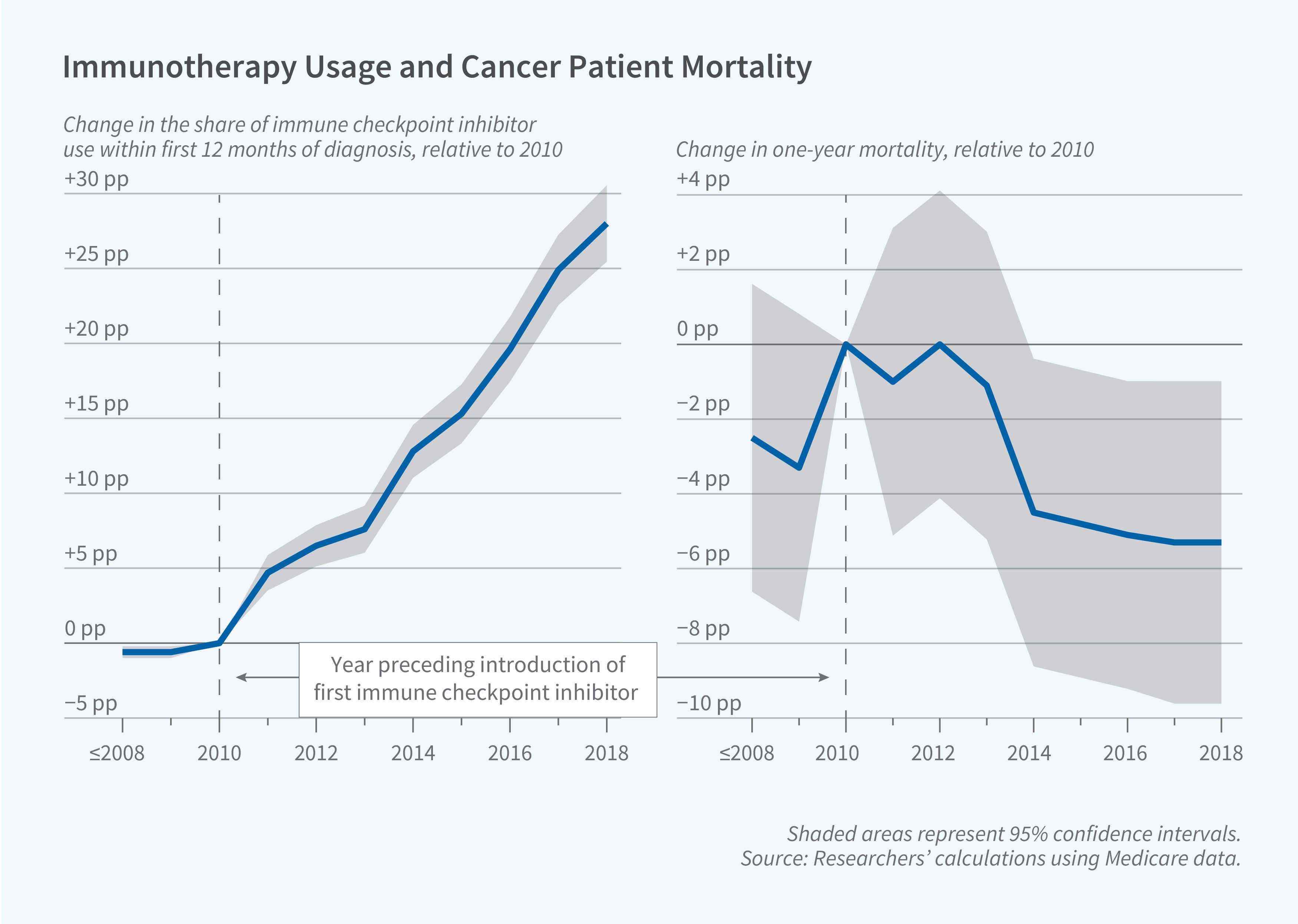
Consistent with earlier approval and broader eligibility standards for treatment of melanoma, the utilization of ICIs increased more for melanoma patients. The use of ICIs among melanoma patients rose from 5.4 percent in 2011 to 33.7 percent in 2018; utilization among colorectal cancer patients was only 5.0 percent in 2018. The researchers estimate that the overall take-up of ICIs increased 22.5 percentage points more among melanoma patients than among colorectal cancer patients after the approval of all three ICIs.
The introduction of the first three ICIs was also associated with a $19,000 differential rise in Medicare costs per melanoma patient, a 59.3 percent increase over baseline spending. The increased spending was concentrated in outpatient care, the setting in which ICIs are administered, with some offsetting declines in expenditures on inpatient and hospice care. The implied increase in total treatment costs for the subset of patients who used ICIs was $85,000, a 263 percent rise. The researchers also find a decrease in the use of chemotherapy and radiation due to the introduction of ICIs.
Further, the introduction of ICIs initiated a striking differential decline in mortality rates for melanoma patients. After all three ICIs were introduced, one-year mortality for metastatic melanoma patients fell by 3.6 percentage points (6.2 percent). The implied impact on the patients who adopted ICIs was a 13 percentage point (28 percent) reduction in one-year mortality.
The researchers conclude that benefits from increased life expectancy for melanoma patients are comparable to, or plausibly greater than, the considerable additional Medicare costs.
- Robin McKnight
The researchers acknowledge support from National Institute on Aging grant #5P01-AG005842-32, National Human Genome Research Institute grant #R01-HG011792, and the Emerson Collective.


