Tighter Prescription Regulations Limited the Rise of Opioid Use
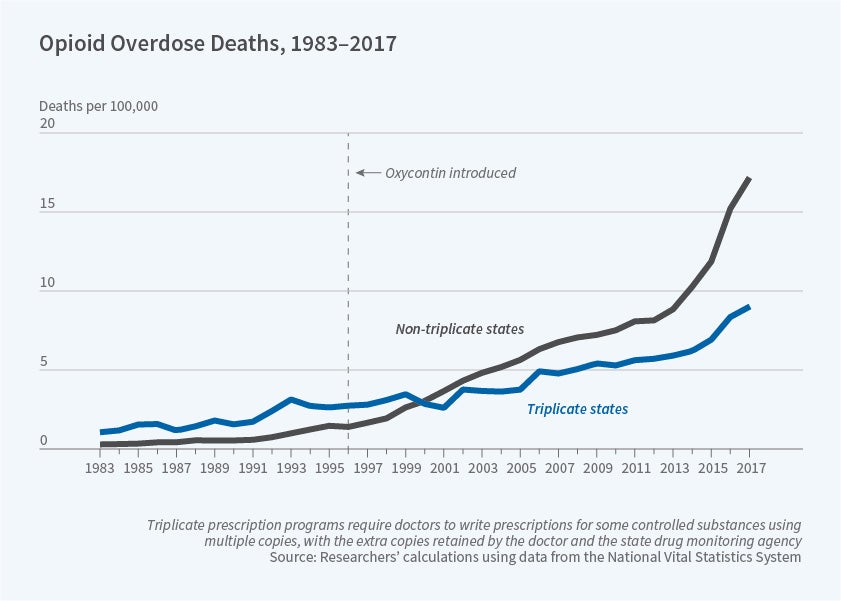
Five states that required doctors to fill out triplicate forms and report opioid prescriptions experienced slower growth in OxyContin use.
The opioid epidemic struck with different force in different states. The average per capita usage rate over the last two decades of OxyContin, a widely used prescription opioid that was introduced in 1996, was about 50 percent lower in California, Idaho, Illinois, New York, and Texas than in other states. In Origins of the Opioid Crisis and Its Enduring Impacts (NBER Working Paper 26500), Abby E. Alpert, William N. Evans, Ethan M.J. Lieber, and David Powell show how regulations in these states influenced the marketing strategy of Purdue Pharma, the maker of OxyContin.
The researchers conclude that what separated the five states which experienced less OxyContin use from the pack was “paperwork.” In 1996, the year the blockbuster painkiller came on the market, these states had the most stringent opioid regulations in the nation, known as triplicate prescription programs. Doctors were required to fill out triplicate prescription forms, sending a copy to the state drug monitoring agency and keeping one on file. While OxyContin prescriptions were lower in the triplicate states, use of other painkillers that were not subject to stringent regulations was just as high in triplicate states as in other states.
According to internal company communications that were revealed in court documents, Purdue Pharma decided against mounting a significant marketing effort in the triplicate states after learning from focus groups that providers would be less willing to prescribe OxyContin in these states. To disentangle the impact of the triplicate rules themselves from the reduced marketing of OxyContin, the researchers compared the five triplicate states with two other groups of states that had had similarly stringent prescribing cultures: five non-triplicate states that had the lowest oxycodone prescribing rates in 1991–95; and two states that had dropped triplicate regulations just prior to OxyContin’s introduction. In all non-triplicate states as well as both sets of comparison states with similar prescribing cultures, OxyContin use and opioid overdose deaths were substantially higher on average than in the five states that had triplicate policies in place when the drug came on the market.
By 2004, all the triplicate states had dropped the triplicate regulations in favor of electronic record-keeping. However, Purdue appears to have continued to concentrate its marketing efforts elsewhere. Internal documents show that the company pushed its sales force to target the top OxyContin prescribers, thus reinforcing prior geographical trends. In 2013–16, total per capita payments to physicians for meals, travel and gifts linked to OxyContin promotion were between 42 and 72 percent higher in the non-triplicate states than in the five states that had triplicate policies in 1996.
The researchers conclude that the cross-state disparities in marketing efforts for OxyContin that originated in the era of triplicate prescriptions help to explain why residents of some states were shielded from the worst of the opioid crisis. Over the 1996–2017 period, non-triplicate states would have had an average of 36 percent fewer drug overdose deaths and 44 percent fewer opioid deaths if they had been triplicate states and experienced the concomitant lower level of marketing. This evidence suggests that the introduction and marketing of OxyContin played a significant role in the beginnings of the opioid crisis and continue to affect overdose rates even today.